As part of National Science Week 2019, we are highlighting some of the high-impact research being carried out by AINSE scholars. In the Materials Science and Engineering area, Monash University student and AINSE PGRA scholar Thomas McCoy is developing new methods to control atomically-thin materials with applications in water purification and oil recovery.

Controlling atomically-thin carbon in water with detergents
Thomas M. McCoy, Liliana de Campo, Anna V. Sokolova, Stephen A. Holt, Rico F. Tabor
Since the discovery of the atomically-thin carbon material ‘graphene’ over a decade ago, research into two-dimensional materials has surged with considerable efforts being devoted to discovering new 2D substrates and ways of utilising their unique properties.
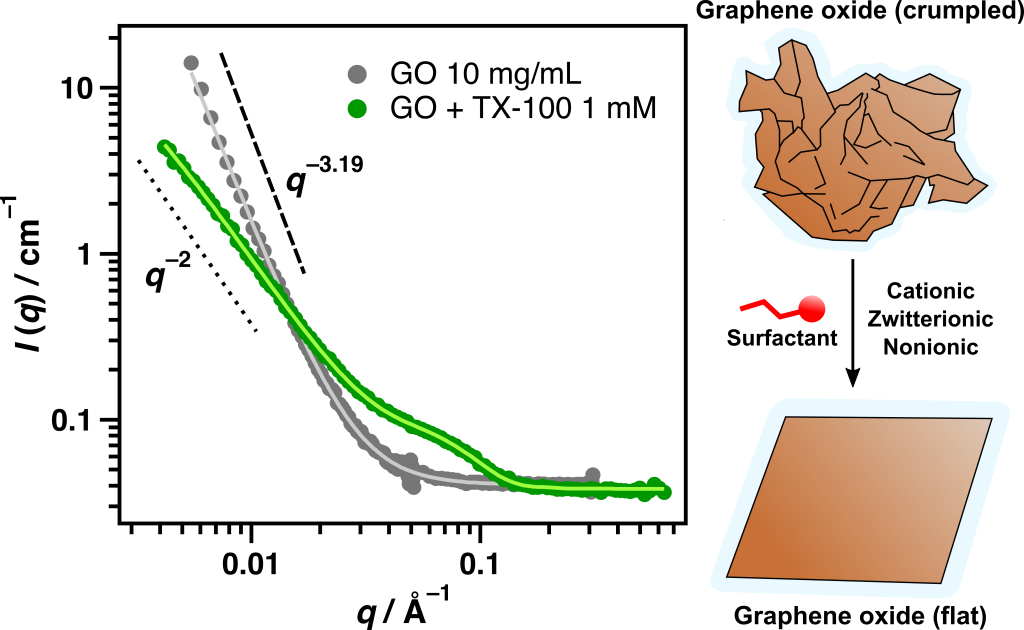
Of particular interest is the water-dispersible analogue of graphene called graphene oxide (GO), a robust material that, while only a single nanometre thick, can range up to tens of microns in diameter (Figure 1). This gives the material an ultrathin sheet-like structure, and due to a high degree of oxygen functionality it can easily be dispersed in water.

These properties make GO an extremely promising material for environmental applications such as water purification and oil recovery, where high surface area is a huge advantage. The challenging aspect of working with these materials is controlling their behaviour when dispersed in water. The ability to do this would not only improve the effectiveness of the materials during application, but also allow them to be recovered once used, which is essential in environmental applications.
“…these novel systems could also be used for enhanced oil recovery with stimulus responsive release capabilities.”
Our work aims to develop and understand methods by which graphene oxide can be controlled in aqueous solution using small molecule adsorbates called surfactants (or detergents). Depending on the chemistry of these compounds, the surfactant molecules can stick to the surfaces of the GO sheets and cause them to aggregate or redisperse, allowing for their capture and reuse (McCoy et al. 2018).
Interestingly, small-angle neutron scattering (SANS) experiments conducted at Lucas Heights have also indicated that the surfactants change the morphology of the GO sheets in solution from crumpled to flat, indicated by the change in slope of the scattering pattern at low scattering vector q (Figure 2) (McCoy et al. 2018). We have dubbed this phenomenon `nano-ironing’, which could be useful in coating and nanotemplating applications.
In addition to affecting the dispersion stability of the GO sheets, surfactant adsorption on the sheets was also found to greatly increase their affinity for interfaces such as the air–water and oil–water interfaces. X-ray reflectivity measurements performed at ANSTO suggest that the GO sheets and surfactant molecules act in synergy for enhanced adsorption at the interface (Figure 3a,c) (McCoy et al. 2017).
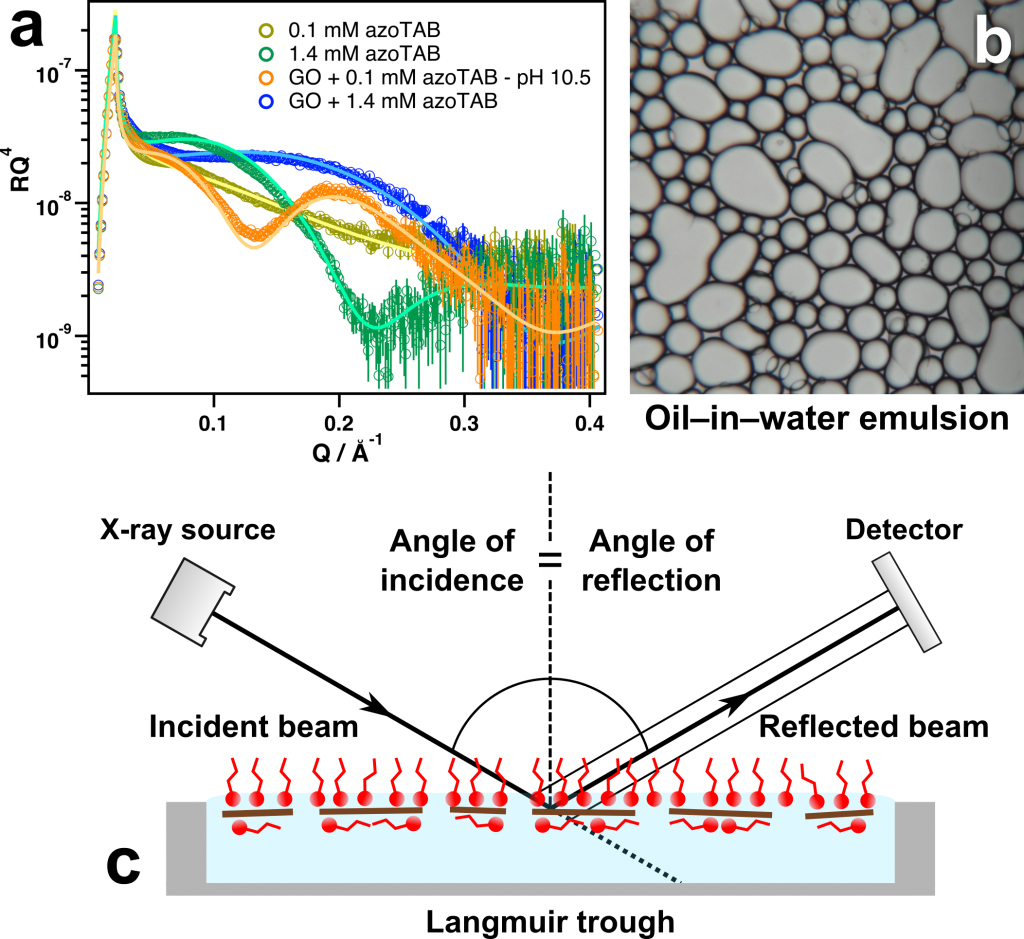
The particular surfactant molecule used in this system was also sensitive to different wavelengths of light that significantly alter its surface, allowing this effect to be manipulated using light as a clean and low energy external stimulus (McCoy et al. 2017). These results suggest that these novel systems could also be used for enhanced oil recovery with stimulus responsive release capabilities.
Acknowledgements:
We would like to acknowledge support from the Australian Institute of Nuclear Science and Engineering, as well as the Monash Centre for Atomically Thin Materials for sponsoring this work. The outcomes of this work would not have been possible without the incredible support of ANSTO beamline scientists Liliana de Campo (SANS/USANS), Anna Sokolova (SANS) and Stephen Holt (XRR). Their diligence and expertise ensured that the experiments were carried out meaningfully and successfully.
References:
McCoy, T M, de Campo, L, Sokolova, A V, Grillo, I, Izgorodina, E I & Tabor, R F 2018, ‘Bulk properties of aqueous graphene oxide and reduced graphene oxide with surfactants and polymers: adsorption and stability’, Physical Chemistry Chemical Physics, vol. 20), pp. 16801–16816.
McCoy, T M, Holt, S A, Rozario, A M, Bell, T D M & Tabor, R F 2017, ‘Surfactant-enhanced adsorption of graphene oxide for improved emulsification of oil in water’, Advanced Materials Interfaces, vol. 4, p. 1700803.
