Glioblastoma is one of the most aggressive and difficult-to-treat forms of brain cancer. Despite advances in surgery, chemotherapy, and radiation therapy, prognosis remains poor, highlighting the urgent need for more precise and effective treatment strategies. Targeted radionuclide therapies (TRTs) offer a promising new approach by delivering highly potent radiation directly to cancer cells while minimising damage to surrounding healthy brain tissue.
Harnessing Meitner–Auger electrons
Meitner–Auger electrons (MAEs) are a form of radiation that travel extremely short distances (10–100 nm) and cause significant biological damage precisely where they are deposited. The use of MAEs in the field of TRTs could open new avenues for the treatment of glioblastoma while sparing precious healthy brain tissue. For these strategies to be effective, however, MAE-emitting isotopes must be delivered specifically to the nucleus of cancerous cells, where they can inflict maximal DNA damage.
Targeting DNA repair pathways
PARP-1 is an enzyme responsible for DNA repair and is overexpressed in many cancers, including glioblastoma. Commercially available and FDA-approved PARP-1 inhibitors, such as Olaparib, target this enzyme located within the nucleus of cancerous cells. Developing strategies to link MAE emitters to PARP-1 inhibitors may therefore enable precise delivery of these isotopes directly to the cell nucleus. The emerging isotope ¹⁹⁷mHg can emit up to 35 MAEs per decay cycle, making it a promising candidate for this purpose.
Developing a targeted radiopharmaceutical
AINSE PGRA recipient Meaghan Ashton (pictured), supervised by Prof. Hugh Harris at the University of Adelaide, successfully conjugated (chemically linked) mercury chelators she developed to the PARP-1 inhibitor Olaparib. Mercury chelators are specially designed molecules that tightly bind to mercury atoms, holding them securely so they can be safely carried and delivered to a specific target — such as cancer cells.
Under the supervision of ANSTO researchers A/Prof Ben Fraser and Dr Flora Mansour, Meaghan used specialised radiolabelling facilities at ANSTO, Lucas Heights, to securely attach the radioactive surrogate isotope ²⁰³Hg to her compound. This process resulted in a radiochemical yield exceeding 99%, indicating that nearly all of the radioactive material was successfully incorporated.

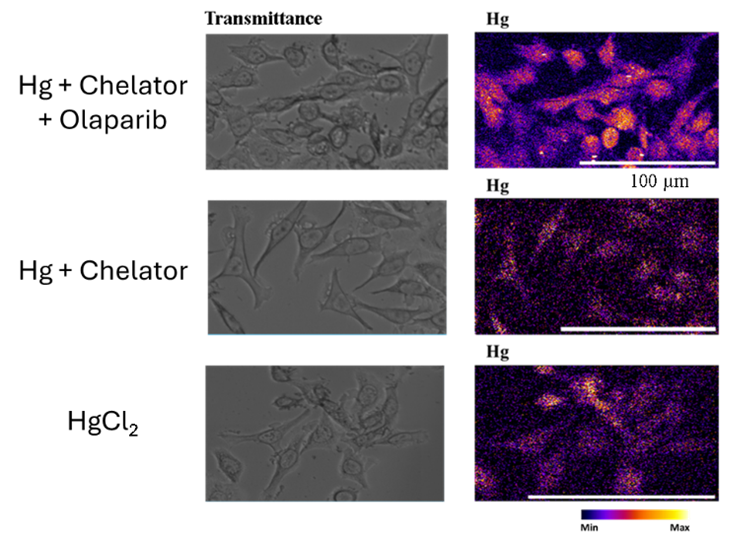
In collaboration with Dr Veronika Pape, cancer cells were treated with non-radioactive versions of these complexes and imaged using the X-Ray Fluorescence Microscopy beamline at the Australian Synchrotron in Melbourne. These experiments provided clear evidence of cellular uptake and localisation of mercury within the nucleus when bound to the Olaparib targeting vector. This nuclear localisation was not observed when mercury was bound to the chelator alone or administered as a free mercury salt.

These results demonstrate strong potential for the use of ¹⁹⁷mHg conjugated to PARP-1 inhibitors as a highly localised, short-range weapon in the fight against glioblastoma.
Meaghan Ashton’s research is an exciting development in the continued fight against aggressive and often terminal brain cancers. By combining the precision of PARP-1 inhibitors with the potent, short-range effects of Meitner–Auger electrons, ¹⁹⁷mHg conjugates offer a highly targeted approach that maximises damage to cancer cells while minimising harm to healthy tissue. These findings lay the groundwork for further development of nuclear-targeted radiopharmaceuticals and represent an exciting step toward more effective therapies for glioblastoma.
Want to get involved?
If you, just like Meaghan, are interested in conducting cutting-edge research in nuclear science with ANSTO, visit https://www.ainse.edu.au/scholarships/ to explore AINSE scholarships.
While you’re waiting for the next Research Spotlight, check out https://www.ainse.edu.au/research-spotlights/ to see the incredible work of past scholars.
